Evolve Your QC Strategy—Without Abandoning Proven Science.
FREE Live Event
Date to be determined (August or September 2026)
Risk Management MasterClass
Build on Proven QC Science to Achieve Risk-Based Clinical Excellence

Extending Proven QC into Today’s
Risk-Based Laboratory
Statistical quality control transformed laboratory medicine by helping detect analytical error. Today’s laboratories face additional operational complexity, regulatory expectations, and patient-risk demands. This masterclass shows how to extend proven QC methodologies with real-time risk intelligence aligned with modern global standards.
Who Is This For?
Laboratory Professionals
Medical laboratory scientists, technologists, supervisors, and managers looking to master risk management
Quality Leaders
QA/QC coordinators, laboratory directors, and pathologists responsible for quality and patient safety
Forward-Thinking Leaders
Anyone seeking to implement value-based laboratory medicine and quantify ROI of quality improvements
Why Do You Need This?
RiskGATOR creates a win-win-win for everyone who cares about laboratory quality
Developed by clinical QC experts. Aligned with CLSI EP23A, ISO 15189, and modern risk-based QC principles.

Lower Healthcare Costs
Unnecessary QC tests, repeated analyses from false alarms, and lab errors that lead to wrong treatments all waste healthcare dollars that could be used to help more patients. Traditional QC estimates costs but doesn’t optimize them.
RiskGATOR quantifies avoidable healthcare costs caused by lab errors, evaluates the acceptability of error costs, and recommends improvements to reduce expenses. It guides labs to create QC processes verified to detect failure in a single run—eliminating waste.
Result: Healthcare systems save money on QC materials, staff time, and patient care—redirecting resources where they’re needed most.

Happier Lab Professionals
Lab professionals spend countless hours investigating false-positive QC flags, repeating tests, and dealing with processes that assume “QC processes are effective” without verification. This creates stress, burnout, and wasted time on problems that don’t actually threaten patient safety.
RiskGATOR reduces false positive QC flags and guides you to focus on real risks. It analyzes QC data to advise if an analytical process actually needs improvement in method accuracy or precision—not just arbitrary sigma values.
Result: Lab professionals reclaim hours each week, reduce stress, and focus on meaningful quality improvements.

Healthier Patients
Every lab error that reaches a patient can delay treatment, lead to incorrect diagnoses, or cause unnecessary procedures. Traditional QC methods remain essential for detecting analytical variation, but statistical indicators alone cannot always quantify patient impact in real time.
RiskGATOR quantifies the actual probability of harm—estimating how often an analytical process could produce one error every ‘x’ hours, days, weeks, months, or years. This means fewer missed errors, more accurate results, and better patient outcomes.
Result: Patients receive safer, more reliable lab results that lead to correct treatment decisions.
AWEsome Numbers’ RiskGATOR empowers laboratory staff to work smarter, more efficiently, and faster by turning complex statistics into clear insights. Our innovative software and proprietary algorithms help standardize quality control, reduce errors, and ensure every result is accurate and reliable.
Exclusive Bonuses for Attendees
Core Questions Answered
The Clinical Impact
- How do lab errors directly create patient harm and increase costs?
The Data Gap
- Which analytical shifts matter most to patient risk?
The Evolution
- How can risk management strengthen existing QC strategies?
The Standards
- What do ISO 15189:2022, CLSI/CLIA, and EFLM recommend?
Acceptable Limits
- How do you define Total Allowable Error that protects patients?
Risk Metrics
- Likelihood of errors vs Sigma metrics: what matters most?
Exclusive Bonuses for Attendees
Interactive Simulator
Change risk drivers for analytical quality and QC effectiveness. Watch real-time impact on patient risk and healthcare costs. Gain a deeper understanding of your data and what affects outcomes.
Paradigm Shift Thinking
Gain New Perspectives
- Learn from global experts about regional challenges with accreditation, QC practice, and lab stress.
- Case studies of reductions in patient errors and lab false positives
- Reduce false positives and simplify lab processes
Interactive Simulator Access
30-day complimentary access to Interactive Risk Simulator to model how QC works and forecast patient risk
Full simulator access
Practice scenarios
Unlimited attempts
Special Pricing
Exclusive discounted pricing on RiskGATOR software
Early adopter pricing
Exclusive discounts
Limited-time offer
50% Discount for 3-hour Online Risk Management Course
Live question & answer time
Video Replays
Free Downloads:
Best Practice Checklist for Quality Control & Risk Management
QC Problem-Solving Worksheet for Root Cause Analysis & Action Planning
Interactive Simulator
Take the blindfold off sigma! Convert sigma to Medically Incorrect Results (MIRs) per year to see how:
-
- variations in acceptable risk impacts Margin for Error (that drives QC process design) while sigma remains constant
- the risk of errors varies with patient volumes while sigma remains constant
- acceptability of risk varies with the TEa value selected,
- Margin for Error and the risk of errors vary with analytical bias, while sigma remains constant
- Margin for Error and the risk of errors vary with method SD
- software can report the acceptability of risk instead of just statistics that you need to interpret
- software can report risk metrics of the number, percent and probability of errors, instead of just statistics
- Margin for Error (the number of SD to unacceptable risk) varies with acceptable risk and patient volume, while sigma remains constant
- Risk Distribution Graphs that show current and failed performance adds meaning add clarity to QC data interpretation
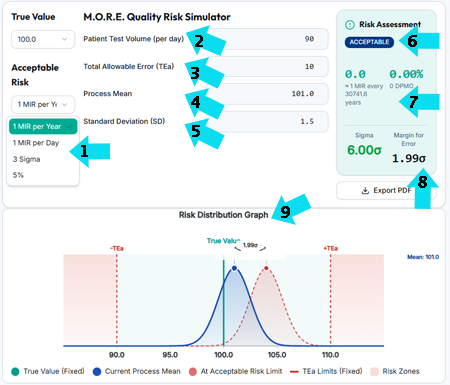
See how risk-based analytics complement statistical QC by translating analytical variation into operational and patient-impact decisions.
Interactive Simulator
Take the blindfold off sigma! Convert sigma to Medically Incorrect Results (MIRs) per year to see how:
-
- variations in acceptable risk impacts Margin for Error (that drives QC process design) while sigma remains constant
- the risk of errors varies with patient volumes while sigma remains constant
- acceptability of risk varies with the TEa value selected,
- Margin for Error and the risk of errors vary with analytical bias, while sigma remains constant
- Margin for Error and the risk of errors vary with method SD
- software can report the acceptability of risk instead of just statistics that you need to interpret
- software can report risk metrics of the number, percent and probability of errors, instead of just statistics
- Margin for Error (the number of SD to unacceptable risk) varies with acceptable risk and patient volume, while sigma remains constant
- Risk Distribution Graphs that show current and failed performance adds meaning add clarity to QC data interpretation
The Assumptions Behind Today’s QC Decisions
Recognize any of these?
The PT Fallacy
- “My proficiency testing is good, so quality is acceptable.”
The Detection Myth
- “I believe our current rules detect failure immediately.”
The Sigma Trap
- “QC rules and frequency can be based only on sigma.”
The SD Error
- “You should combine several reagent lots to establish method SD.”
The Global Perspective
Learn how ISO 15189:2022, CLSI/CLIA, and EFLM regulations are driving the shift toward Risk Management worldwide.
USA | Canada | Europe| India | Asia | Latin America | Africa
What You Gain
-
-
-
-
- Reduce False Positives:
Stop wasting time chasing false alarms. - Staff Empowerment:
Make quality/risk management easier to understand and execute. - Patient-Risk Visibility:
See how analytical variation translates into medically incorrect results before patient harm occurs. - Operational Decision Intelligence:
Turn sigma, bias, SD, and TEa data into clear action thresholds your team can execute with confidence. - Smarter QC Scheduling:
Learn when additional QC adds protection—and when it only adds cost, delay, and alarm fatigue. - AI in the Lab:
Learn how AI improves just-in-time understanding and guidance. - Accreditation Confidence:
Document QC decisions with risk-based evidence aligned with ISO 15189, CLSI, and global best practices. - Confidence to Innovate:
Learn how to strengthen QC decisions using risk-based evidence—without abandoning proven statistical methodologies. - Discount for Certification Course: Self-paced deep-dive Risk Management training program.
- Reduce False Positives:
-
-
-
Join the Evolution of Quality Control
Extend proven QC science with real-time operational risk intelligence.
Upgrade your quality systems, reduce stress, improve patient safety, and lead the next generation of laboratory excellence.
Your Instructor and Moderator
Zoe Brooks
Co-Founder & CEO, AWEsome Numbers Inc
Building on decades of statistical QC science, Zoe developed the M.O.R.E. (Mathematically-OptimiZed Risk Evaluation) methodology to connect analytical variation with operational and patient-risk decision making.
Zoe is a globally recognized authority on laboratory risk management and quality control. Author of “Performance-Driven Quality Control” (AACC Press), multiple articles and scientific posters including the Award Winning “Impact of Seven Incremental Scenarios of QC Strategies. Her M.O.R.E. methodology powers AWEsome Numbers’ RiskGATOR software. (see more)

What happens next?
We’ll send you event details, access links, and information about your exclusive bonuses!

